|
This lone pair pushes the S-F bonds away, much like in bent or trigonal pyramidal geometries. This allows the fluorine atoms to be the greatest distance apart from one another considering there is a lone electron pair on the central sulfur atom. 
The other two S-F bonds direct away from each other in 3-dimensional space. In an SF 4 molecule, two of the S-F bonds are situated across from one another in the equatorial plane. SF 4 is an example of a molecule with a seesaw shape. In the ammonia molecule, the lone pair on the central nitrogen atom pushes the three N-H bonds downwards due to electron-electron repulsion. NH 3 is an example of a trigonal pyramidal molecule. The bond angle between the hydrogen atoms is 104 degrees. In a water molecule, the lone pairs on the oxygen atom force the hydrogen bonds downwards in 2-dimensional space. When the central atom in a molecule has lone pairs, these lone pairs repel the bonds rooted in the central atom. VSEPR Theory of molecules with lone electron pairs on the central atom Bent The repulsion between the fluorine atoms can be minimized by placing each one at the corner of an octahedron. Each S-F bond accounts for two valence electrons, and each fluorine atom carries three lone pairs. There are 48 valence electrons in an SF 6 molecule. SF 6 is an example of an octahedral molecule. In order for the equatorial fluorines to be the furthest distance away from each other as possible, the P-F bonds are at a 120-degree angle with respect to each other, and the bond angles between the equatorial and axial positions are 90 degrees. The other two are in what is called the axial position. Three of the fluorine atoms are in what is called the equatorial position. Each P-F bond uses 2 valence electrons and each fluorine atom has three lone pairs. PF 5 is an example of a Trigonal Bipyramidal molecule. The H-C-H bond angle in a tetrahedral molecule is 109.5 degrees. Tetrahedral is the 3-dimensional expression of square planar geometry. In order for the four hydrogens to be furthest apart from one another, we arrive at a tetrahedral shape. There are eight total valence electrons in a methane molecule. TetrahedralĪ common example of a tetrahedral molecule is CH 4 (methane). CO 3 (carbonate) is another example of a trigonal planar molecule. The bond angles in a trigonal planar molecule are 120 degrees. In molecular geometry, this is known as “trigonal planar”. In order for each fluorine atom to keep its lone pairs as far away as possible from the other fluorine atoms, the molecule forms a triangular, 2-dimensional shape. There are 24 total valence electrons in a BF 3 molecule. Trigonal PlanarīF 3 is an example of a trigonal planar molecule. CO 2 and BeH 2 are also linear molecules. The bond angles in a linear molecule are 180 degrees. 
In order for these lone pairs on each respective fluorine atom to be the furthest distance possible from the other fluorine atom, the molecule forms a straight line. There are 16 total valence electrons in a BeF 2 molecule, with three lone pairs (six electrons)on each fluorine atom.  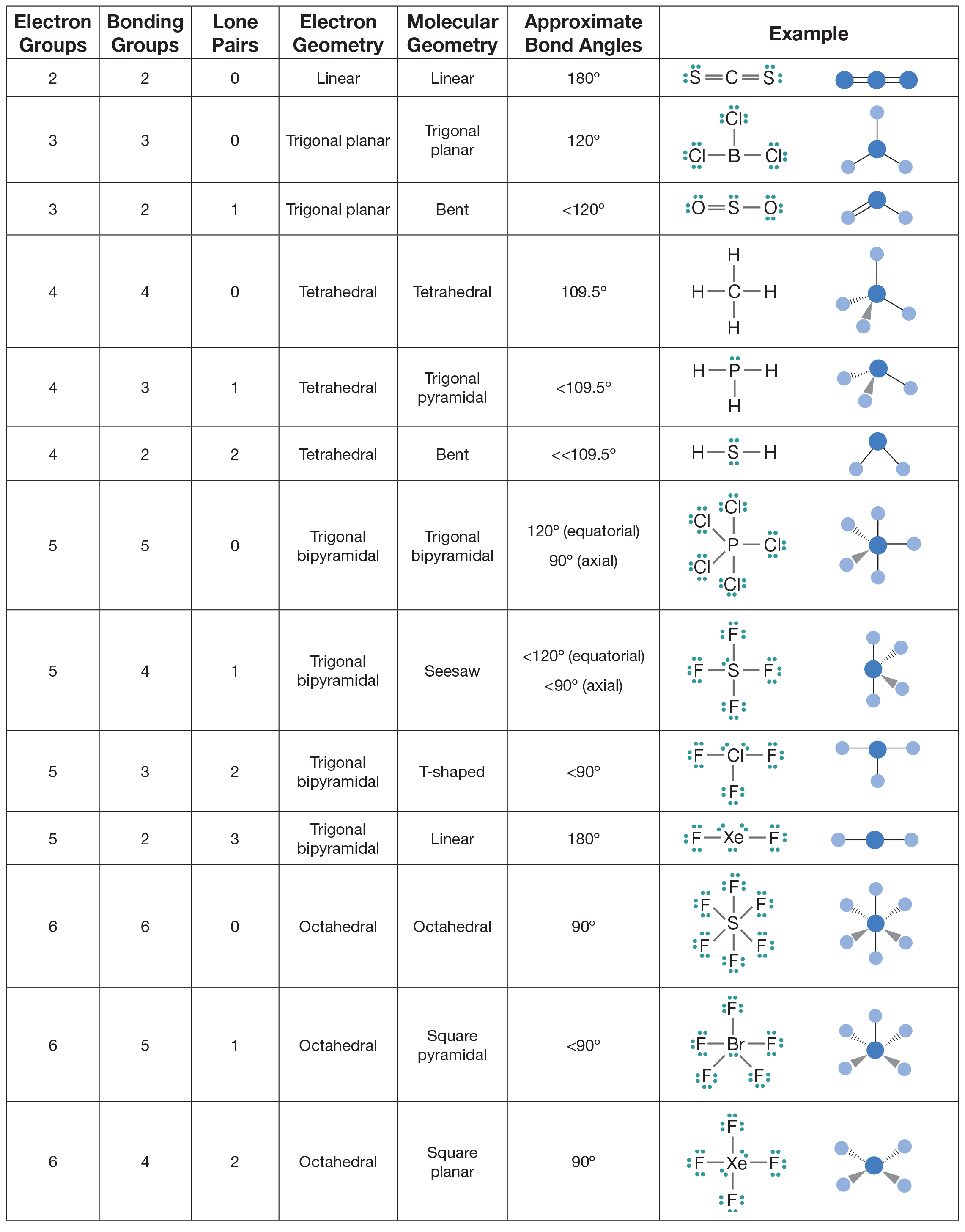
LinearīeF 2 is an example of a linear molecule. VSEPR Theory of Molecules without lone electron pairs on the central atomįor simplicity and organization, we will separate VSEPR structures into two categories: those with lone pairs on the central atom, and those without lone pairs on the central atom. Valence electrons repel one another because they are negatively charged and like charges repel. In chemistry, VSEPR Theory is based on the principle that each atom in a molecule will seek a geometry that maximizes the distance between valence electron pairs, thus minimizing electron-electron repulsion. VSEPR Theory is short for Valence Shell Electron Pair Repulsion Theory, a method of organizing molecules based on their geometric structures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
